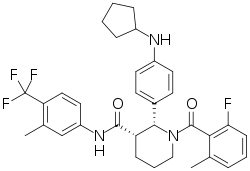
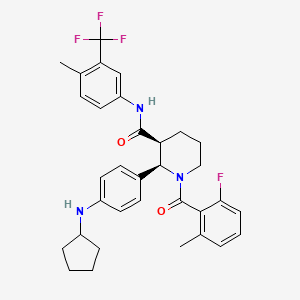
VFMCRP and ChemoCentryx announce positive topline data from pivotal phase- III ADVOCATE trial demonstrating avacopan's superio

ChemoCentryx's Tavneos (avacopan) Receives the US FDA's Approval as an Adjunctive Treatment for Adult Patients with

VFMCRP and ChemoCentryx announce European Medicines Agency has accepted to review the Marketing Authorization Application for avacopan | Business Wire